Developmental and genetic regulation of the human cortex transcriptome illuminate schizophrenia pathogenesis
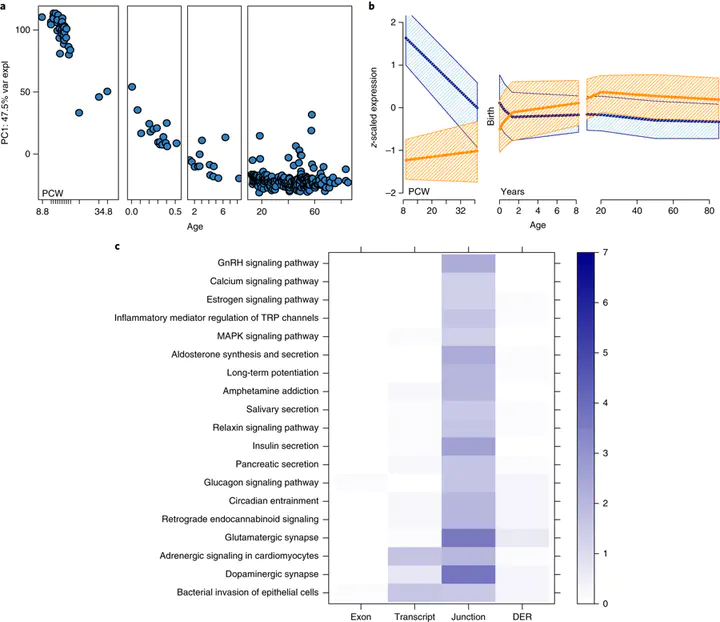 Image credit: Nature Neuroscience
Image credit: Nature Neuroscience
Abstract
Genome-wide association studies have identified 108 schizophrenia risk loci, but biological mechanisms for individual loci are largely unknown. Using developmental, genetic and illness-based RNA sequencing expression analysis in human brain, we characterized the human brain transcriptome around these loci and found enrichment for developmentally regulated genes with novel examples of shifting isoform usage across pre- and postnatal life. We found widespread expression quantitative trait loci (eQTLs), including many with transcript specificity and previously unannotated sequence that were independently replicated. We leveraged this general eQTL database to show that 48.1% of risk variants for schizophrenia associate with nearby expression. We lastly found 237 genes significantly differentially expressed between patients and controls, which replicated in an independent dataset, implicated synaptic processes, and were strongly regulated in early development. These findings together offer genetics- and diagnosis-related targets for better modeling of schizophrenia risk. This resource is publicly available at http://eqtl.brainseq.org/phase1.
Check out our most recent @LieberInstitute work on eQTLs, DLPFC development and #schizophrenia at https://t.co/CN8QO1xGlV lead by @andrewejaffe. Thanks to @biorxivpreprint for letting us share our progress https://t.co/6Zds7FW4U7 and @NatureNeuro for publishing the final version! pic.twitter.com/CKJxoXmAdu
— 🇲🇽 Leonardo Collado-Torres (@lcolladotor) July 26, 2018
Most comprehensive eQTL resource in human brain to date, finds half of risk snps for schizophrenia map to nearby expression, including unannotated sequence https://t.co/Q4TGxChb66 @LieberInstitute check out browser at https://t.co/PdoshZZE5J
— Andrew Jaffe (@andrewejaffe) July 26, 2018